You can think of it as corrosion-if you get it in contact with anything, it corrodes everything.” “The difficulty is, lithium metal is too reactive. Chief among them, says Liu, is its reactivity. To tap lithium’s potential, researchers have spent decades working through the metal’s numerous roadblocks. 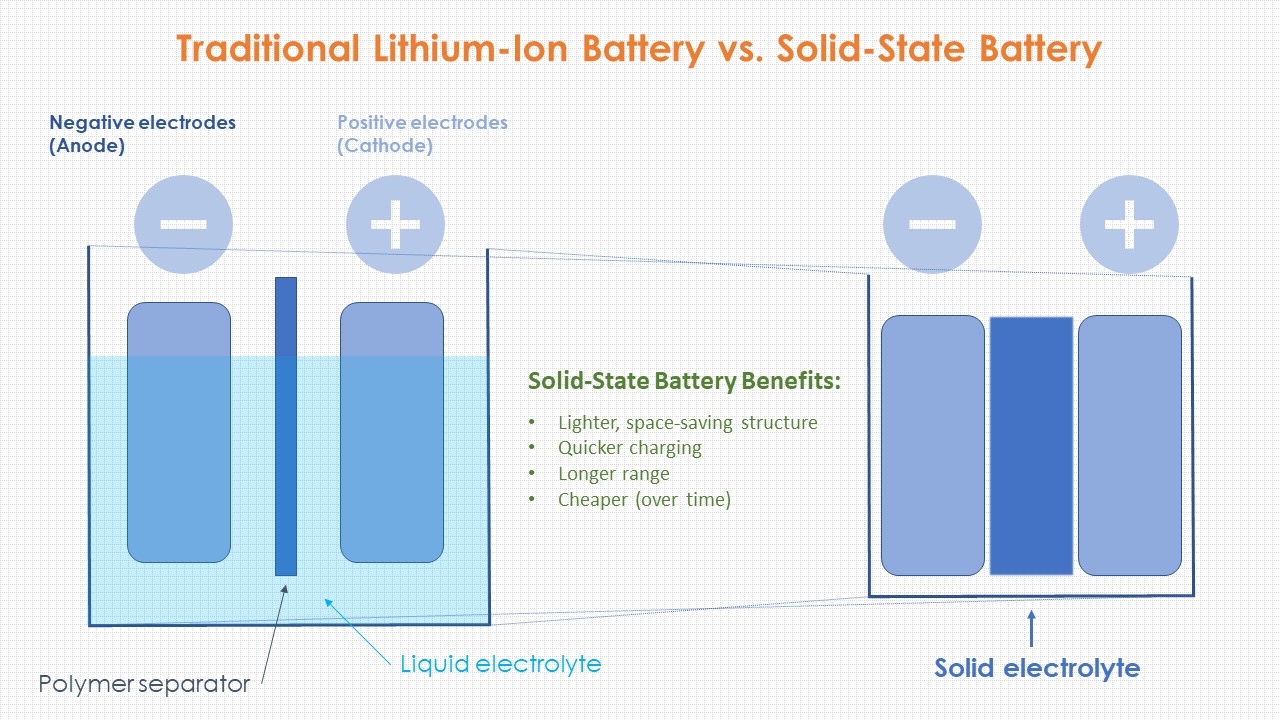
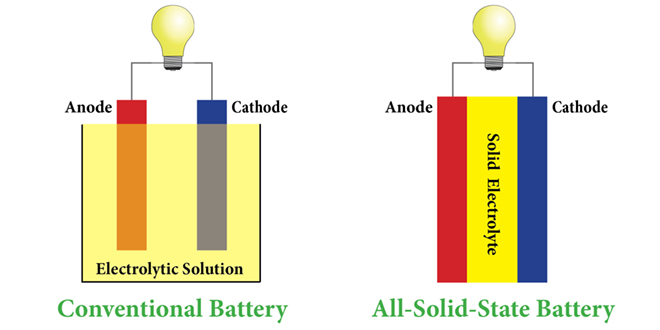
However, lithium metal’s capacity is 10 times that of graphite. This material is plentiful, conducts well, and is easy to work with. The cathode (positive electrode) is a lithium compound, and the anode (negative electrode)-which determines total storage-is made of graphite. Current lithium-ion batteries use a liquid electrolyte where ions flow back and forth between the anode and cathode, recharging and discharging electrons (see How Lithium-Ion Batteries Work, below). The energy density of lithium-ion cells is as much as four times greater than that of the nickel-cadmium batteries they’ve largely replaced. And compared with other alkalis, such as potassium or sodium, lithium has the smallest ion size-and third-lowest atomic weight on the periodic table-meaning more electrons and charge for a given battery size. “That creates a really high voltage,” he explains. Like its fellow alkali metals on the far left of the periodic table, lithium has a single outer electron that it easily gives up, says Jeff Sakamoto, Ph.D., a mechanical engineering professor at University of Michigan who specializes in solid-state battery research. Lithium has been the focus of battery research for decades because it’s an excellent conductor. Sakamoto and his team created a battery with double the output of lithium ion. He felt confident, but a little apprehensive: “We knew we could make something that looked like a battery cell, but there was still a chance we’d have a brick.” Josh Buettner-Garrett, Solid Power’s chief technology officer, monitored from his office. Then on August 7, 2021, three engineers donned protective Tyvek “bunny suits,” entered the dry room, and drew voltage from the largest prototype lithium-metal battery to date. 
Solid Power was aiming for more modest gains in its first prototypes, but could still see an 80 percent improvement in the near future. The technology, in theory, sounded too good to be true: a 10x jump in power (or 10x drop in size) from traditional lithium-ion cells. The cells, a shining silver contrast to their surroundings, were a moonshot. It’s here in the humidity- and contaminant-free production area where Solid Power produced their first full-size solid-state lithium-metal battery cells. However, the higher interface resistance and higher-cost, lower-yield manufacturing processes show some difficulties in general.The dry room at Solid Power’s Louisville, Colorado, facility is abrasively bright, and yet the low, encompassing hum of the fans and chillers is oddly soothing. Oxide systems are more stable than lithium metal, with good electrochemical and thermal stabilities. While the relatively high operating temperature, low anti-oxide potential and worse stability indicate challenges. Polymer systems are easy to fabricate, most compatible with existing manufacturing facilities, and some are already commercialized. However, the difficulty of manufacturing and the toxic by-product hydrogen sulfide generated in the process make the commercialization relatively slow. Many features make them appealing, being considered by many as the ultimate option. In general, sulfide electrolytes have the advantages of high ionic conductivity, even better than liquid electrolyte, low processing temperature, wide electrochemical stability window, etc. Oxide, sulfide and polymer systems have become the most popular options in the next-generation development, with further variations under each category. Within the solid-state battery regime, there are various technology approaches. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
